Crack-cocaine dependence and aging: effects on working memory

Print version ISSN 1516-4446 – On-line version ISSN 1809-452X
Rev. Bras. Psiquiatr. vol.38 no.1 São Paulo Jan./Mar. 2016
http://dx.doi.org/10.1590/1516-4446-2015-1708
Brief Communications
Crack-cocaine dependence and aging: effects on working memory
1Developmental Cognitive Neuroscience Research Group (GNCD), Centre of Studies and Research in Traumatic Stress (NEPTE), Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, RS, Brazil
Revista Brasileira de Psiquiatria
Objective:
To compare the working memory (WM) performance of young adult crack-cocaine dependent users, healthy older adults, and a control group of healthy young adults.
Methods:
A total of 77 female participants took part in this study: 26 young adult crack-cocaine dependent users (CRK), 19 healthy older adults (HO), and 32 healthy younger adults (HC). All participants completed the N-back verbal task.
Results:
A multivariate analysis of covariance was performed. The model included education, income, and medication use as covariates. A group effect (F6,140 = 7.192, p < 0.001) was found. Post-hoc analyses showed that the performance of the CRK and HO groups was reduced compared to the HC group in two N-back conditions. No differences between the HO and CRK groups on WM performance were found.
Conclusions:
CRK participants perform similar to HO participants on a WM task, despite the well-known effects of age on WM and the young age of CRK. These data point to a possible parallel between cognitive declines associated with crack use and developmental aging.
Key words: Working memory; crack cocaine; aging; substance use-related disorders; cognition
Introduction
Crack-cocaine use has been shown to cause toxic effects on the brain, particularly in the prefrontal cortex (PFC). Such abnormalities are associated with neuropsychological impairments, including deficits in working memory (WM)., Interestingly, PFC alterations and decline in WM performance are recognized as normal consequences of natural aging. Imaging data supports the hypothesis that gray matter volume loss over time is twice as fast among cocaine users as in healthy individuals. Given that gray matter volume in PFC has been related to WM performance, it is presumed that cocaine use impacts WM as well, which has been corroborated by behavioral results. In addition, preliminary data suggest that cocaine use and aging have interactive effects on neuropsychological integrity, increasing impairments and everyday problems.
WM is a high-demand cognitive process that involves maintaining and manipulating information in the absence of external cues. WM has been described as critical to several other cognitive processes, such as executive functioning and social cognition, as well as to everyday functioning. Deficits in WM performance are also associated with clinical symptoms found among cocaine users, such as higher impulsivity traits and higher dosages of drug consumed.
Given the suggested importance of WM to the neuropsychological functioning of cocaine users and the hypothesis that cocaine use could cause a decline in WM performance, this study sought to compare the WM of young adult crack-cocaine users to that of healthy older adults and healthy young adults. The hypothesis is that adult crack users would exhibit WM performance equivalent to that of the older group instead of their age-controlled healthy peers.
Method
Participants
Since crack-cocaine use and cognitive aging show gender effects, only women were included in this study. Seventy-seven women were recruited and selected by convenience. The sample was separated into three groups: healthy adult controls (HC, n=32); healthy older participants (HO, n=19); and crack-cocaine dependent users (CRK, n=26). The age cutoffs used to determine the participants as adults or older adults were based on the criteria established by the World Health Organization (adults, 19-59 years; older adults, > 60 years). The two groups of young adult participants (HC and CRK) were age-controlled to avoid age-related biases. The exclusion criteria were as follows: history of neurological illness, head injury, current pregnancy, dementia symptoms as assessed by the Brazilian version of the Mini-Mental State Examination (MMSE), current treatment for any substance or alcohol dependence (controls only), and any psychoactive drug use (except caffeine and prescribed drugs) within 24 h prior to testing. The Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders-IV (SCID-I) was used to determine exclusion/inclusion of healthy participants.
All healthy participants (HC and HO) were recruited through institutional advertisements and assessed by a clinical interview. The CRK group was recruited from a public inpatient detoxification program. All participants from the CRK group fulfilled the DSM-IV criteria for cocaine substance dependence, as confirmed by the SCID-I. Patients had abstained from drugs or alcohol for at least 14 days at the time of cognitive evaluation (mean = 17.30±0.55 days). Among the CRK group, the mean age at alcohol use onset was 14.97±5.35 years; the mean age at cocaine/crack use onset was 18.32±8.4 years; and the lifetime duration of cocaine/crack use was around 7.67±9.87 years. Some participants in the CRK group fulfilled criteria for depression (n=7), post-traumatic stress disorder (n=3), alcohol use/dependence (n=8), and cannabis use disorder (n=4). All CRK participants reported smoked cocaine (i.e., crack cocaine) use.
Instruments and procedures
To assess WM performance, the Brazilian version of a verbal N-back task described by Dobbs & Rule was used. In the task, digits are presented at a rate of one per second. Unlike in the digit-span task, rather than remembering a list of numbers, the subject must identify when the current stimulus matches a stimulus n steps back in the sequence. The N-back task was selected because it is a popular WM test. As its loading increases, more and more cognitive effort is demanded, making it sensitive enough to detect subtle WM differences between groups. In this study, three different target conditions, n=1, n=2, and n=3, were used. Before each procedure, a study phase was conducted to make sure each participant understood the rules. The participant should remember the number at n steps back in the list following the presentation of each further item. There was a total of 10 items to be remembered in each condition and it is assumed that, the more the participant can remember, the better the WM performance.
Data analyses
Data were screened for violations of normality and logarithmic transformations were performed when necessary. Demographic variables were compared using one-way analysis of variance (ANOVA). Additionally, N-backs 1, 2, and 3 were used in a multivariate analysis of covariance (MANCOVA) as dependent variables. The group was included as a fixed factor and all individual variables that had shown significant group differences were considered as covariates in the MANCOVA model except age, which took part in the group variable. Post-hoc tests based on Tukey’s test were performed to examine pairwise comparisons. The significance level was set at α < 0.05 (two-tailed).
Results
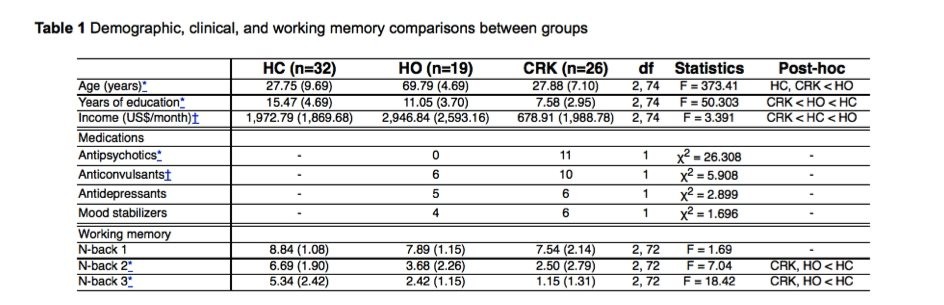
The groups differed in years of formal education, individual income, use of antipsychotics, and use of antidepressants, and these were included as covariates in the MANCOVA model. The group was set as the fixed factor and the measures of the N-back 1, 2, and 3 were the dependent variables. A significant group effect was found (F6,140 = 7.192, p < 0.001). Tukey post-hoc analysis showed that the CRK and HO groups exhibited similar WM performances in all N-back levels, and that both the CRK and HO groups had poorer performances than the HC group in N-back 2 and 3. Table 1 shows the results of comparative analysis.
Table 1 Demographic, clinical, and working memory comparisons between groups
| HC (n=32) | HO (n=19) | CRK (n=26) | df | Statistics | Post-hoc | |
|---|---|---|---|---|---|---|
| Age (years)* | 27.75 (9.69) | 69.79 (4.69) | 27.88 (7.10) | 2, 74 | F = 373.41 | HC, CRK < HO |
| Years of education* | 15.47 (4.69) | 11.05 (3.70) | 7.58 (2.95) | 2, 74 | F = 50.303 | CRK < HO < HC |
| Income (US$/month)† | 1,972.79 (1,869.68) | 2,946.84 (2,593.16) | 678.91 (1,988.78) | 2, 74 | F = 3.391 | CRK < HC < HO |
| Medications | ||||||
| Antipsychotics* | – | 0 | 11 | 1 | χ2 = 26.308 | – |
| Anticonvulsants† | – | 6 | 10 | 1 | χ2 = 5.908 | – |
| Antidepressants | – | 5 | 6 | 1 | χ2 = 2.899 | – |
| Mood stabilizers | – | 4 | 6 | 1 | χ2 = 1.696 | – |
| Working memory | ||||||
| N-back 1 | 8.84 (1.08) | 7.89 (1.15) | 7.54 (2.14) | 2, 72 | F = 1.69 | – |
| N-back 2* | 6.69 (1.90) | 3.68 (2.26) | 2.50 (2.79) | 2, 72 | F = 7.04 | CRK, HO < HC |
| N-back 3* | 5.34 (2.42) | 2.42 (1.15) | 1.15 (1.31) | 2, 72 | F = 18.42 | CRK, HO < HC |
Data presented as mean (standard deviation).
CRK = crack-cocaine dependent users; df = degree of freedom; HC = healthy younger adults; HO = healthy older adults.
Post-hoc analyses were based on Tukey’s test. For working memory variables, results were controlled for years of education, individual income, and medication use.
*p < 0.001;
†p < 0.05.
Discussion
Behavioral WM data indicated a similar performance among crack-cocaine users and healthy older adults, which is poorer than that of healthy younger adults. Such results are in agreement with previous studies of differences in WM performance both of cocaine dependents, and of healthy older adults, vs. typical healthy young adult participants.
Our results add suggestive data to the hypothesis of previous studies that cocaine use may cause a “fast-track” aging process., A previous study found evidence that both cocaine users and healthy older adults show white matter reductions in the hippocampus and PFC compared to healthy younger adults. Since the integrity of gray matter in these brain regions has been related to WM integrity, our results further support these findings. Nevertheless, further detailed investigations – e.g., looking for neurobiological evidence of the origins of cognitive problems within healthy, elderly adults and crack-cocaine users – would be interesting. Heroin has been shown to cause cellular changes similar to those found in aging, and it is possible that these accelerated aging processes contribute to earlier onset of behavioral problems in drug users.
The main limitations of this study include the use of a single measure for the evaluation of WM, lack of a wider neuropsychological investigation, the brief abstinence period of crack-cocaine users, the presence of other psychiatric disorders, and the heterogeneity across groups regarding individual characteristics. General intelligence is an important issue that could account for WM performance and other individual differences among groups. The similar results on N-back for cocaine dependents and healthy older participants may have different etiologies, meaning that differences could be a consequence of different neuropsychological functions, e.g., processing speed and inhibition, and not necessarily of WM impairments. Early abstinence among crack-cocaine users could affect WM performance differently than in other phases of substance use. Finally, the sample was heterogeneous with regard to income, years of formal education, and medication use. As a comprehensive evaluation of individual characteristics was not possible, statistical methods were used to control for such differences.
We cannot state that crack-cocaine dependence ages WM, nor identify the origins of the WM impairments within each condition. However, our findings are relevant, particularly for crack-cocaine dependent patients. To date, treatments for drug use have been planned mainly for young adults, despite increasing numbers of older adults with drug use problems. According to these results, the WM performance of crack-cocaine users does not match the expected WM performance for their age. It is possible that current interventions do not match the capacities of crack users; therefore, studies focusing on WM training combined with motivational interviewing might help improve executive functioning in cocaine dependents. In addition, WM training has been found to help improve inhibitory control in stimulant addicts. In summary, participants with crack-cocaine dependence, despite their younger age, exhibited almost the same WM performance of participants over the age of 60, whereas healthy, young adults performed significantly better.

